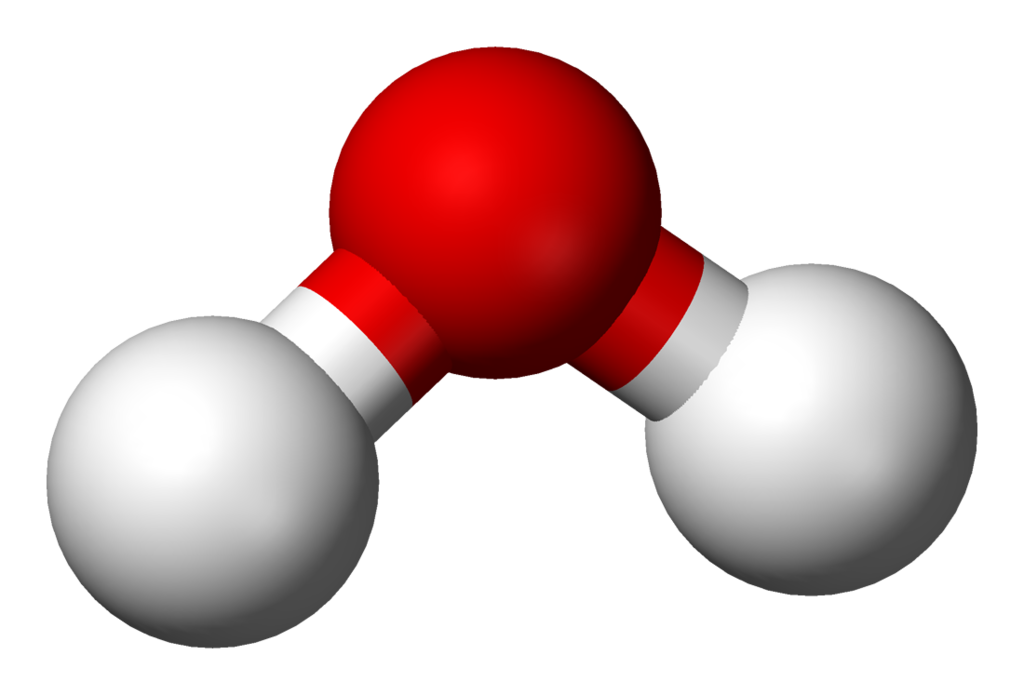
Water
IUPAC: Oxidane

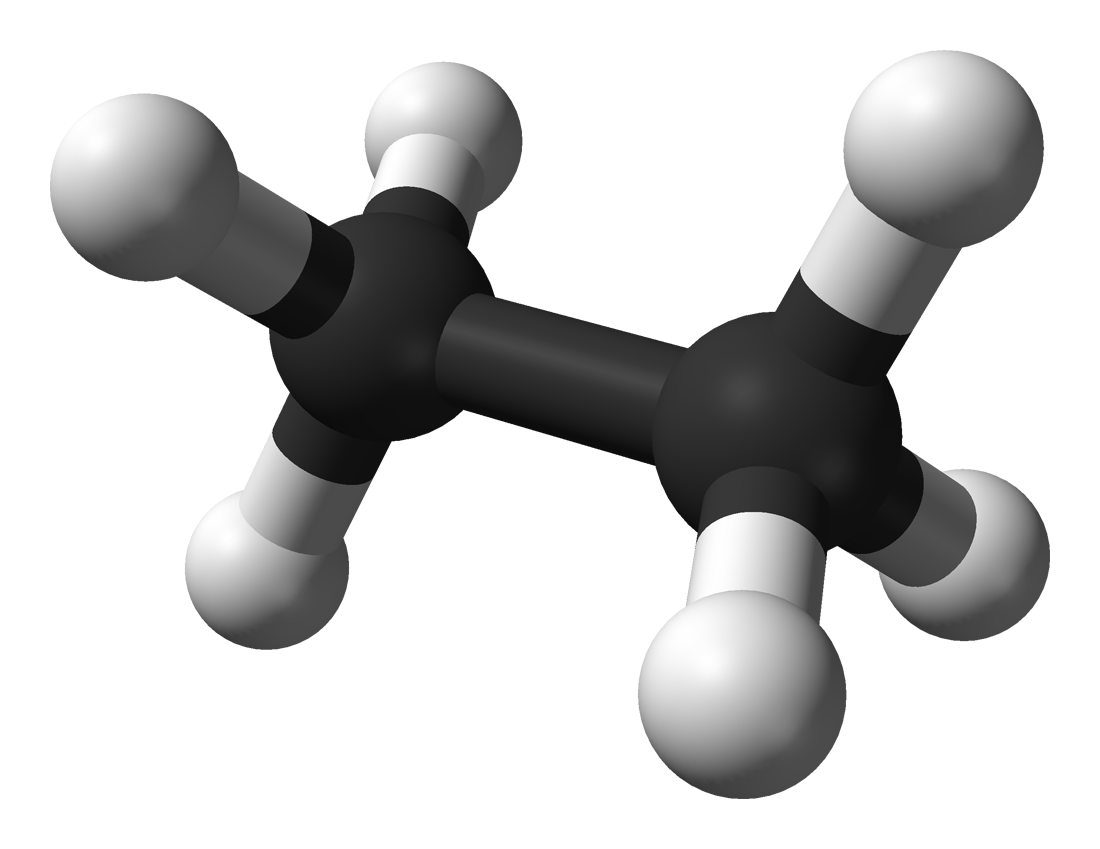
Ethane
IUPAC: Dicarbane

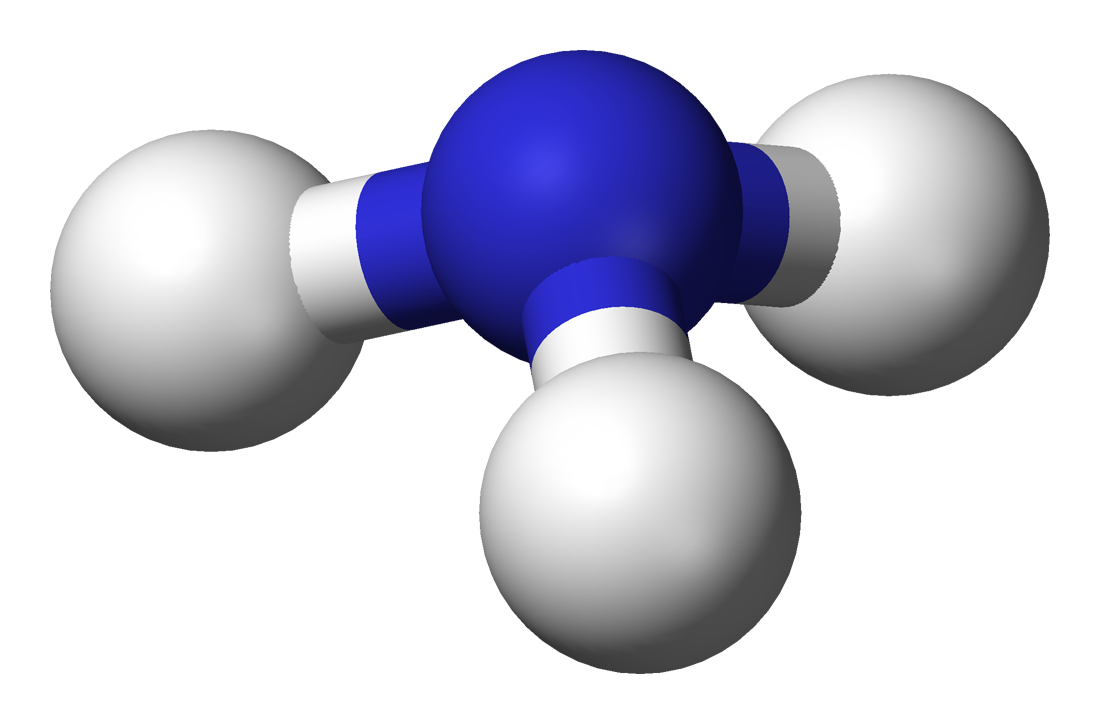
Ammonia
IUPAC: Azane

Questions
- Post an image from the web, the chemical systematic (IUPAC) name, common name, and the molecule formula for 20 chemicals that you use or eat. Perhaps explore the ingredients of things like cosmetics, foods and/or automotive product.
Sodium Chloride -Table Salt – NaCl

Ethyl Alcohol – Mouthwash – CH3CH2OH

Sodium Bicarbonate- Baking Powder- NaHCO3

Acetic acid or Ethanoic acid- Vinegar- C2H4O2

Sucrose- Sugar- C12H22O11

Dichlorobenzene- Moth Balls- C6H4Cl2

Calcium Carbonate- Chalk- CaCO3

Ethanol- Alcohol- C2H6O

Graphite (Carbon)- Black Lead- C

Sodium Hypochlorite- Bleach- NaOCl

Sulphuric Acid- Battery Acid- H2SO4

Acetylsalicylic Acid- Aspirin- C9H8O4

Aluminum Chlorohydrate – Deodorant – Al2Cl(OH)5

Sodium Hydroxide – Drain Cleaner – NaOH

Ethylene Glycol – Antifreeze – C2H4(OH)2

Sodium Carbonate Decahydrate – Water Softner – NaCO3⋅10H2O

Boric Acid- Insecticide- H3BO3

Sodium Tetraborate Dechydrate- Laundry Detergent- Na2[B4O5(OH)4] 8H2O

Acetone- Nail Polish Remover- CH3COCH3

Butane- Lighter Fluid- C4H10

Dihydrogen Dioxide- Peroxide- C2O2

2. Look over your molecules and the bonding characteristics, how many bonds does each of the following elements typically have? Carbon? Hydrogen? Oxygen?
- Hydrogen- 1
- Carbon- 4
- Oxygen- 2
3. What does IUPAC stand for?
- International Union of Pure and Applied Chemistry
4. As you explore ingredients, notice how everything around us is made up of chemicals consisting of atoms bound together into molecules. But what about companies that claim their products are chemical free! How can this be?
- The claim that companies make that their products are “chemical free” is almost impossible because everything is a chemical at the molecular level. If you think about it vinegar is a chemical, kosher salt is a chemical, and baking soda is also a chemical. The reason companies label their products as chemical free is solely a marketing ploy to make themselves look better than competitors. The companies that say they are chemical free are most likely using more natural chemicals instead of chemicals that are formed in laboratories.
5. Also do a little searching on the web and share on your blog how many chemicals are typically found in things like coffee, milk, beer or whiskey?
- Milk- There can be as many as 20 different chemicals found in your milk such as painkillers, antibiotics and growth hormones. Cows milk contains the highest levels of chemicals when tested with goats milk and breastmilk.
| Anti-inflammatories (niflumic acid, mefenamic acid, ketoprofen, diclofenac, phenylbutazone, naproxen, flunixin, diclofenac) | Antibiotics (florfenicol) | Natural hormones (estrone) |
| Sex hormones (17-beta-estradiol) | Steroid hormones (17-alpha-ethinylestradiol) | Anti-malaria drugs (pyrimethamine) |
| Anti-fungal drugs (triclosan) |
