Neon

Lithium

Lithium Excited

Boron

Questions
What is the atomic number for each of your models?
- Neon- 2
- Lithium- 3
- Boron- 5
What is the atomic mass for each of your models?
- Neon- 20.1797 u
- Lithium- 6.941 u
- Boron- 10.811 u
In your models, which two subatomic particles are equal in number?
- Protons & Electrons, in Neon, Protons, Electrons and Neutrons
How would you make an isotope for one of your models? What would change with the model?
- Neon: To make this an isotope you either take a way an electron or put in another electron so that you either have more or less neutrons than protons.
Considering the overall volume of your element models, what makes up most of the volume of an atom?
- Empty space in which negatively charged electrons move around the nucleus.
Once the electron is excited, what do we typically observe when the electron returns to the ground-state?
- We observe that they are discharged, they lose electrical charge and become neutral. The electrons then flow from the object to the ground.
Why are some elements different colors when they are excited?
- The color of the elements depends on how much energy is being released, and it comes from the different electrons losing their energy.
You may observe fireworks over the New Year’s, explain how the different colors of fireworks arise.
- Red- Strontium carbonate
- Orange- Calcium chloride
- Yellow- Sodium nitrate
- Green- Barium chloride
- Blue- Copper chloride
- Purple- Strontium & Copper compounds
Explain the overall organizational structure of the periodic table.
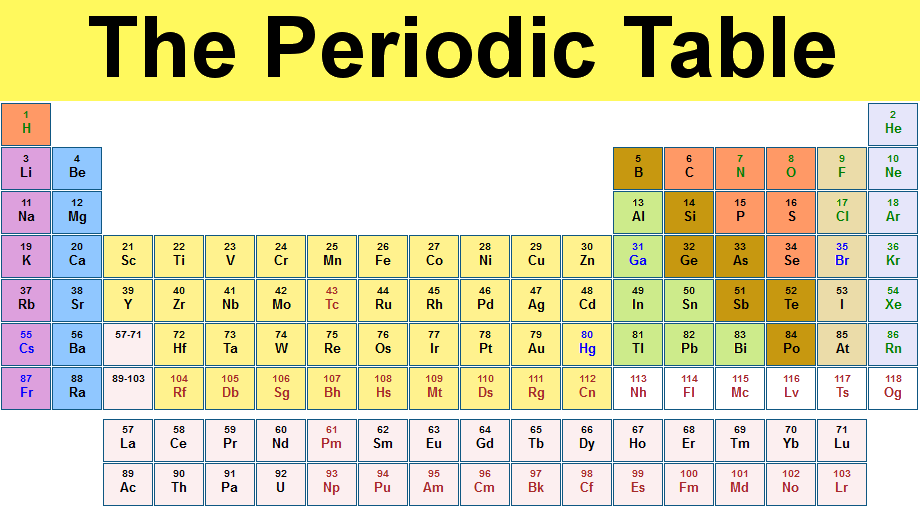
- The periodic table is a chart that lists the elements in order of increasing atomic number. The vertical columns are called groups and they contain elements with similar properties. The horizontal rows are called periods, the atomic number increases in the row from left to right. There are 18 groups and 7 periods in the periodic table.
List two example elements for each of these groups or classes: Alkali Metals, Alkaline Earth, Halogens, Noble Gases, Transition Metals, Non-Metals, and Metalloids.
- Alkali Metals- Lithium & Sodium
- Alkaline Earth- Magnesium & Calcium
- Halogens- Chlorine & Bromine
- Noble Gases- Helium & Neon
- Transition Metals- Silver & Gold
- Non-metals- Hydrogen & Carbon
- Metalloids- Boron & Silicon
