Question:
- Does hot water or cold water freeze faster?
Hypothesis
If 50 mL of cold water and 50 mL of hot water are placed in a freezer simultaneously than the hot water will freeze faster due to the Mpemba effect which is something I learned about in high school chemistry.
Variables
Independent:
- Temperature of the water
Dependent:
- Time it takes the water to freeze
Controlled:
- uniform containers
- water from the same source
- samples cooled in same freezer
Materials
- freezer
- two cups of equal size
- marker
- water
- graduated cylinder
- thermometer
- timer
- notebook
- pen

Procedure
- Fill 2 cups with 50 mL of water. One will contain hot water (100 degrees C) and one will contain cold water (40 degrees C).
- Use the thermometer to measure the exact starting temp.
- Record the starting temperature.
- Set a timer for 10 minutes.
- Place the two cups in the freezer.
- Start the timer.
- After the 10 minutes has passed record the temperature of the water now.
- Place the cups back in for an additional 10 minutes.
- Repeat step 7.
- Start timer for 5 minutes.
- After the 5 minutes has passed record the temperature of the water.
- Set timer for 1 minute increments and record the temperature until one of your cups has reached 0 degrees Celsius.

Cups in freezer 
50 mL in each cup ready to put in freezer
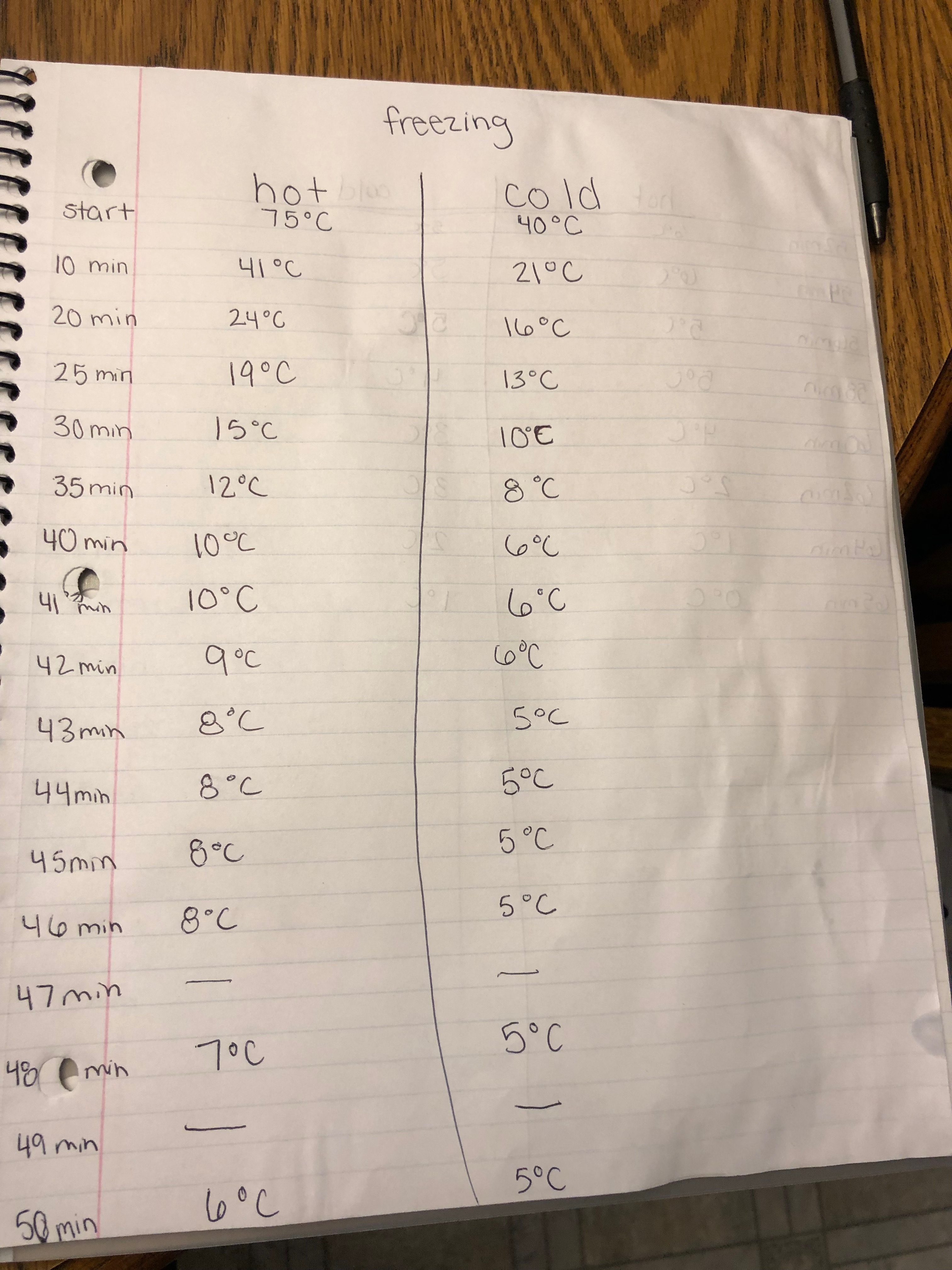
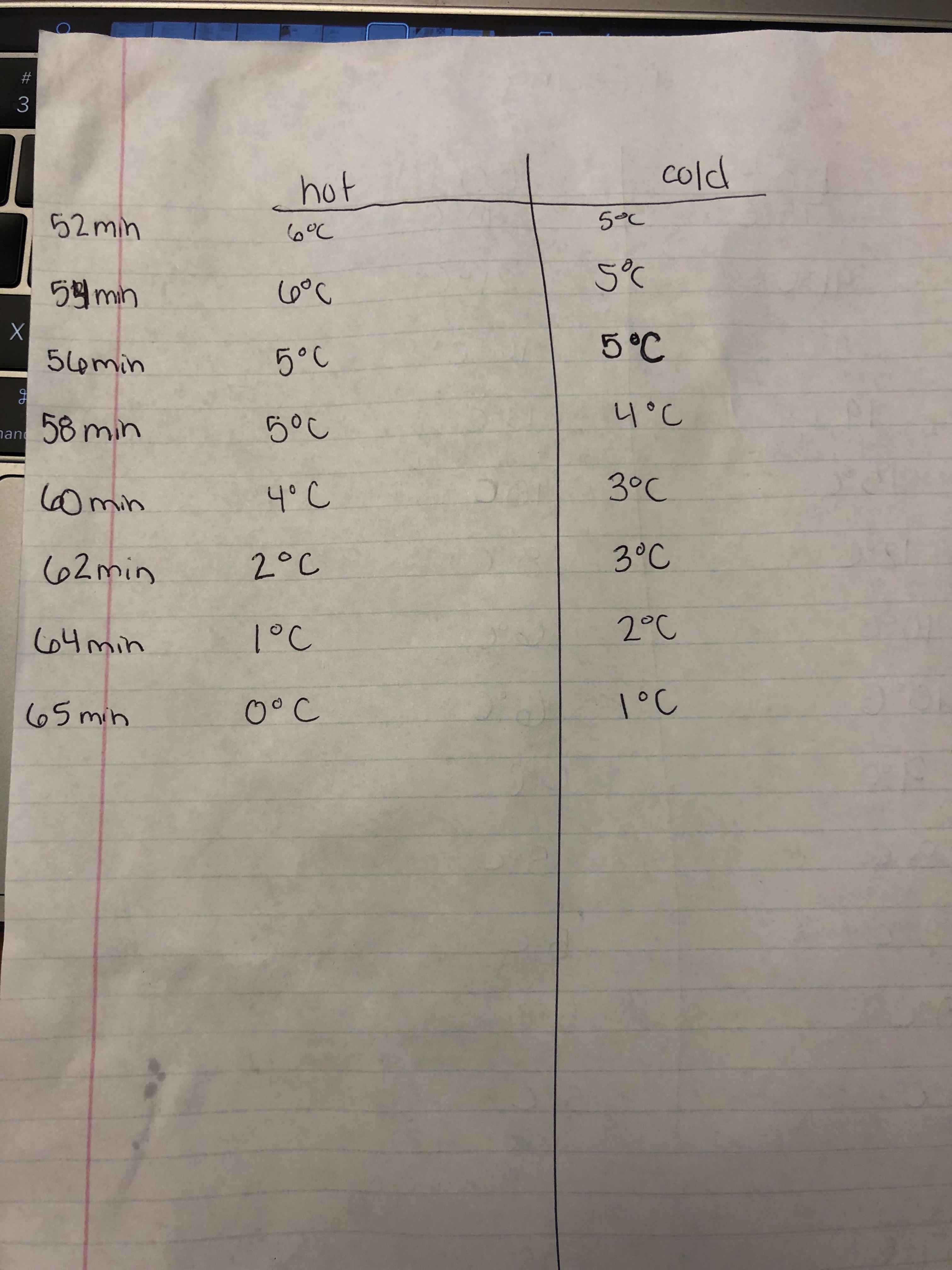
Observations
Data



Theory
In my three trial of my experiment I seen that the hot water froze faster than the cold water, however it was not much faster in each of the three trials. I believe this happened because of a scientific term called the Mpemba Effect. The Mpemba Effect deals with the hydrogen bonds bringing water molecules into close contact. The effect of water molecules being brought into close contact is the O-H bonds stretch and store energy. This is the equivalent to cooling when the bonds give up energy.
Average Values
The average freeze time for hot and cold water was 66 minutes.
Question:
2. Does hot water or cold water boil faster?
Hypothesis
If you boil 2 cups of hot water and 2 cups of cold water, then hot water will boil faster because it is closer to the boiling temperature to begin with.
Variables
Independent:
- Temperature of the water
Dependent:
- Time it takes the water to boil
Controlled Variable:
- Same Pan used for experiment
- Same source of water
- Same source of heat
Materials
- stovetop
- pan w/lid
- measuring cup
- notebook
- thermometer
- pen

Procedure
- Fill pan with 2 cups cold water.
- Take temperature of water.
- Record starting temperature.
- Turn burner on high.
- Start stopwatch at same time you turn the burner on.
- Once water is boiling stop the stop watch and record the end temperature.
- Repeat steps 1-7 with hot water.

Observations
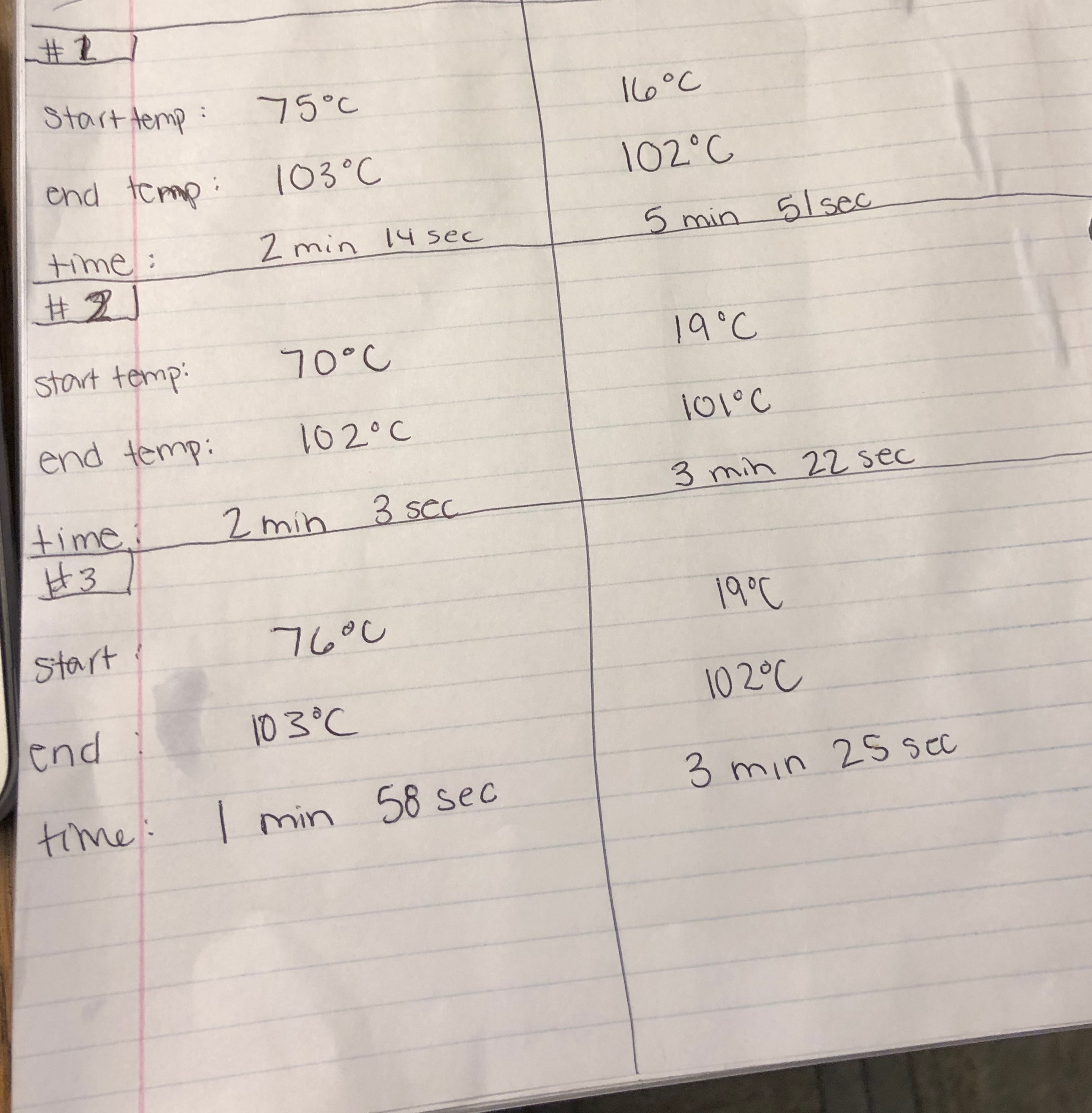
Data



Theory
Although cold water will absorb heat faster until it is to the temperature of hot water, it does not mean that it will boil faster. The hot water was closer to the temperature of the boiling point of water, therefore it boiled faster.
Average Values
The average boil time for hot water was 2 minutes and 8 seconds and the average boil time for cold water was 3 minutes and 32 seconds.
Question
3. Does salt water freeze faster or slower than regular water?
Hypothesis
If you place two cups with 50 mL of water in the freezer, the cup without salt dissolved in will freeze faster because salt water has a lower freezing temperature.
Variables
Independent:
- Amount of salt in water
Dependent:
- Time it takes the water to freeze
Controlled:
- uniform containers
- water from the same source
- samples cooled in same freezer
Materials
- freezer
- two cups of equal size
- marker
- water
- salt
- graduated cylinder
- thermometer
- timer
- notebook
- pen

Procedure
- Fill 2 cups with 50 mL of water. One will contain regular water and one will contain salt water.
- Use the thermometer to measure the exact starting temp.
- Record the starting temperature.
- Set a timer for 30 minutes.
- Place the two cups in the freezer.
- Start the timer.
- After the 30 minutes has passed record the temperature of the water now.
- Place the cups back in for an additional 30 minutes.
- Repeat step 7 until cups are froze over.

Observations

Data



Theory
My theory is regular water freezes faster than salt water because salt is used to melt ice. Also, salt water needs to be colder before it freezes whereas regular water freezes at zero degrees celsius.
Average Values
The average time it took regular water to freeze was between 180 and 210 minutes, at 210 minutes the salt water was beginning to freeze, however it was not completely froze like the regular water was.
Paragraph
Doing this activity we were introduced to a variety of different scientific concepts, the first one was creating and conducting the actual experiment. The science concepts in that were hypothesis, procedure, materials, observations, data and theory. The second science concepts we were introduced too was actually working with water and the three states: solid, liquid, and gas. By doing this we learned how water molecules interact in each state. The last science concept we were introduced to is compounds when we mixed pure water with salt.
When conducting the three experiments I had to set each of them up first, my first step in each one was to create a hypothesis, identify my variables, list my materials needed, and come up with the procedure. These steps were very similar in both the cold water vs hot water and water vs salt water freezing experiments. The only difference in these two experiments was the time between taking the temperature. After completing each of my three experiments I came up with the conclusion that hot water freezes faster than cold water, hot water boils faster than cold water and pure water freezes faster than salt water. For my two freezing experiments I created graphs with my data, and for my boiling experiment I put my data into a table.
All three of these experiments are beneficial for me in the real world because I enjoy cooking and baking. After conducting the experiments I now know that if I need to quickly boil something I will use hot water, same with if I need the water to freeze quicker. Doing these experiments helped me with being efficient in the next time I decide to cook or bake.
Water Molecule